- Blog
- Iron horse bike company
- Adobe premiere pro cs4 green screen
- Persona 5 fusion calculator
- Think of me sheet music free print
- Igri my play city
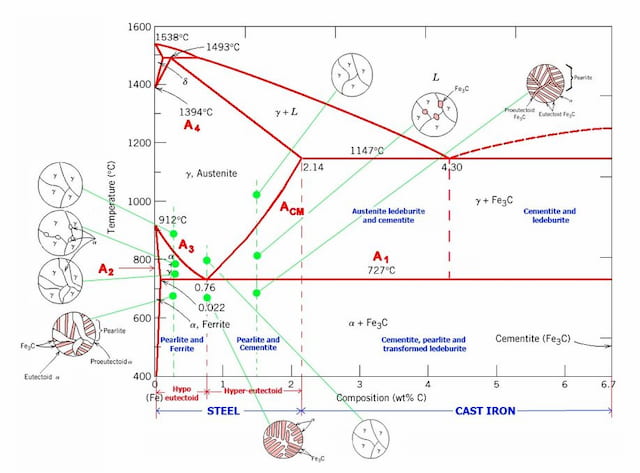
- Binary phase diagram pdf
- Spanish mauser 1916
- I want boardmaker
- Nagisa clannad movie theme
- 96 tamil movie song download
- 2nd and charles
- Asphalt 4 jar dedmoil
- Usb 2-0 driver windows 7
- Install scansnap manager ix500
The position of the point at each end of the tie line, at the boundary of the two-phase area, gives the value of the composition variable of one of the phases and also the physical state of this phase: either the state of an adjacent one-phase area, or the state of a phase of fixed composition when the boundary is a vertical line.
If the system point is in a two-phase area of the phase diagram, we draw a horizontal tie line of constant temperature (on a temperature–composition phase diagram) or constant pressure (on a pressure–composition phase diagram).For example, on a temperature–composition phase diagram, the pressure is fixed and the temperature and composition can be changed independently within the boundaries of the one-phase area of the diagram. On the phase diagram, the value of either \(T\) or \(p\) has been fixed, so there are two other independent intensive variables. If the system point falls within a one-phase area of the phase diagram, the composition variable is the composition of that single phase.The way in which we interpret a two-dimensional phase diagram to obtain the compositions of individual phases depends on the number of phases present in the system. The composition variable usually varies along the horizontal axis and can be the mole fraction, mass fraction, or mass percent of one of the components, as will presently be illustrated by various examples. The position of the system point on one of these diagrams then corresponds to a definite temperature, pressure, and overall composition. Instead of using these variables as the coordinates of a three-dimensional phase diagram, we usually draw a two-dimensional phase diagram that is either a temperature–composition diagram at a fixed pressure or a pressure–composition diagram at a fixed temperature. We can independently vary the temperature, pressure, and composition of the system as a whole. Since \(F\) cannot be negative, the equilibrium system can have no more than four phases. There must be at least one phase, so the maximum possible value of \(F\) is 3.
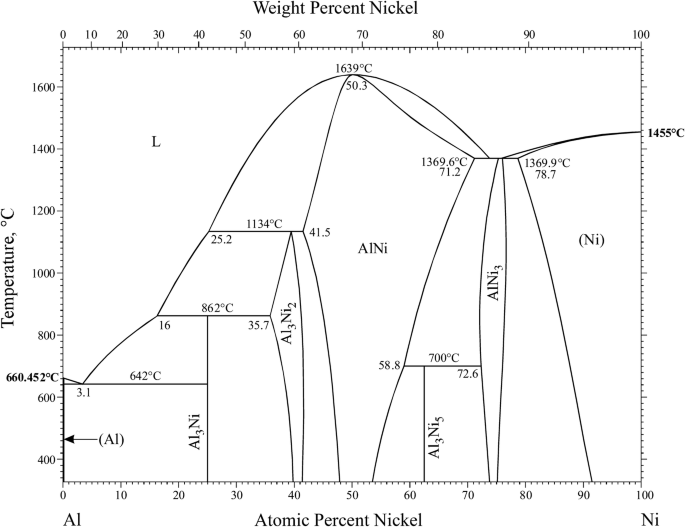

13.3 will describe some interesting ternary systems.Ī binary system has two components \(C\) equals \(2\), and the number of degrees of freedom is \(F=4-P\). This section discusses some common kinds of binary systems, and Sec. 8.2, a phase diagram is a kind of two-dimensional map that shows which phase or phases are stable under a given set of conditions.
